Presence of nutrients, especially nitrogen and phosphorus in wastewater effluents and their impacts on natural water bodies are of major concern. With the recent evidence that anthropogenic phosphorus (not nitrogen) addition in micrograms per liter level can trigger algal growth, phosphorus removal to lowest level has become increasingly important.
Presence of nutrients, especially nitrogen and phosphorus in wastewater effluents cause anthropogenic phosphorus (not nitrogen) addition in micrograms per liter level to trigger algal growth. As a result, phosphorus removal to lowest level has become increasingly important. Chemical and biological means are adopted to remove them. While phosphorus could be removed chemically, nitrogen removal is mostly carried out by biological means. Biological phosphorus removal process is popular over chemical means for it’s simplicity, economy and various environmental benefits.
Biological phosphorus removal process relies on enhancing the ability of microorganisms to uptake more phosphorus into their cell. Therefore, these processes are often referred to as enhanced biological phosphorus removal (EBPR) processes. EBPR has been implemented worldwide in many wastewater treatment plants. Despite its promise to provide efficient phosphorus removal performance, at times unreliable performance has been reported. In an effort to achieve improved phosphorus removal, the addition of more phosphorus removing chemicals, like alkalinity reducing coagulants, can be counterproductive to the Waste Water Treatment Plant.
Three options are available to remove phosphorous from the system: Convert phosphorous to chemical species by adding a metal salt or lime (precipitation); remove with membrane treatments; and incorporate the phosphorous into biomass – the most cost effective is biomass removal. Overall optimization of phosphorus removal can be achieved not through additional changes to the biological system or increasing the use of coagulants to chemically remove it, but rather through improvement the overall ecosystem of the biology throughout the plant. The process is directly dependent on the health of the ecosystem and the availability of critical nutrients and alkalinity. Our research has seen biomass contains 1.5 to 2.5 % (w/w) phosphorous per volatile solids. Under certain conditions, the biomass will accumulate phosphorous levels of 6 to 8 %, far in excess of the nutritional requirements. Phosphorus accumulated in sludge or biomass is removed by sedimentation (solid separation). The process of removing phosphorus by accumulating it with biomass is referred to as enhanced biological phosphorous removal (EBPR) process.
To achieve these higher levels of bioaccumulation, the biomass must be maintained in a stable environment and be augmented to allow for improved phosphorus uptake. One mechanism by which to achieve this is to improve both the alkalinity of the biomass environment, and improve magnesium levels, which is critical for the biology treatment process.
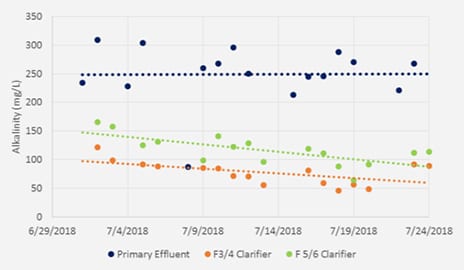
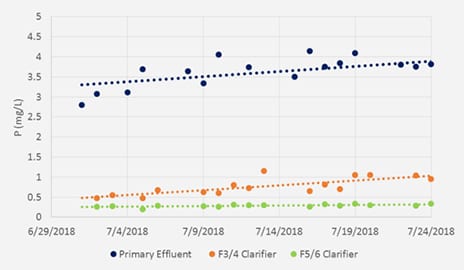
pHREADY is a total system treatment solution that uniquely provides both non-hazardous, stable delivery of alkalinity to a WWTP, but also is rich in ions required for EBPR. By using pHREADY, WWTP’s have seem dramatic improvements in stability in the treatment process but also enhanced phosphorus removal (Figure 1). This unique testing site utilized a split plant design whereby influent could be sent to two isolated treatment plants and evaluated directly against each other. The plant alkalinity (Figure 2) was noticeably improved for the pHREADY side of the plant and provides yielding a boost of approximately 50% improvements. Furthermore Phosphorus removal in the final clarification step (Figure 3) was dramatically improved. Overall during the trial the plant efficiency for removal was improved by 64% without addition coagulation steps or any difference to the treatment process.
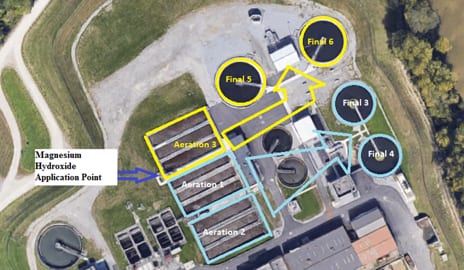
Figure 1. (right) Side by side treatment plants, only Aeration 3 and Final 5 and 6 received treatment with pHREADY. A1/2 and F3/4 were untreated for a control. Incoming influent was split between the two plants.
Figure 2. (below, left) Alkalinity (mg/L) for the Primary process effluent and the Final Clarifiers effluent.
Figure 3. (below, right) Alkalinity (mg/L) for the Primary process effluent and the Final Clarifiers effluent.
As mentioned above, the three mechanisms for phosphorus removal have been deployed for years. The most cost effective for plants is to optimized their biomass removal process as much as possible and augment with chemical precipitation methods. The main drawback for chemical coagulation methods is typically the plant is limited by the total amount of chemical they are able to feed. This is where pHREADY, a total system treatment solution, uniquely provides our customers a pathway to improve further their ability to remove phosphorus. pHREADY is both non-hazardous and stable delivery mechanism for adding alkalinity to a WWTP, but also is rich in ions required for enhanced phosphorus biological removal. With 50% improvements in alkalinity and 64% improvements in Phosphorus uptake, the total sustained health of the biomass is improved and ultimately over time continued to maintain stable removal of phosphorus.
pHREADY is an effective alkalinity booster designed specifically to work in conjunction with SulfAWAY and BioREADY products to treat odor at the source!