Reading Time | 8 Minutes

Most wastewater treatment problems are not chemistry failures — they are chemistry mismatches. A facility manager orders a flocculant when they actually need a coagulant, runs the wrong polymer charge for their solids, or doses both when only one is required. The result is wasted spend, poor settling, and discharge numbers that look worse than they should. Here is how to make the right call
Send us a sample — we'll run the analysis and translate it into plain-language recommendations for your treatment program.
A coagulant is a chemical that neutralizes the electrical charges that keep tiny suspended particles apart in wastewater. A flocculant is a long-chain polymer that bridges those neutralized particles together into larger clumps — called flocs — that are heavy enough to settle, float, or filter out cleanly. Coagulants destabilize. Flocculants aggregate.
That is the entire mechanism. But which one you need — or whether you need both — depends entirely on what your wastewater actually contains. Here is the side-by-side reference that most operators need on the wall:
| Coagulants | Flocculants | |
|---|---|---|
| Job | Neutralize particle surface charge so particles can approach each other. | Bridge and bind those particles into large, settleable flocs. |
| Chemistry | Inorganic salts (alum, ferric chloride, PAC) or organic polymers (PolyDADMACs, polyamines). | Long-chain polymers — typically polyacrylamide-based — in cationic, anionic, or non-ionic form. |
| Mixing | Fast, energetic mix to disperse the coagulant rapidly across the stream. | Slow, gentle mix to grow flocs without shearing them apart. |
| Typical Dose | 10–500 mg/L for inorganic types; 5–50 mg/L for organic coagulants. | 0.5–10 mg/L (parts per million) — very low mass dose. |
| pH Range | Alum: 6.5–7.5 optimal. Ferric: 5.0–8.5. Organic: broad range. | Effective across wide pH ranges; charge type must match particle charge. |
| Sequence | Always added FIRST. | Added AFTER coagulation is complete. |
Download our one-page Coagulant & Flocculant Selection Guide — built from real facility data, it matches your solids type, particle charge, pH, and discharge target to the right chemistry before you call a vendor.
Three questions decide what your wastewater needs. Walk through them in order:
If your wastewater carries coarse, heavy solids that are already destabilized — like aggregate wash water, concrete slurry, or mineral fines from processing — the particles don’t need charge neutralization. They need binding. That’s a flocculant-only application.
Common flocculant-only applications: aggregate and stone wash water, concrete slurry recycling, sludge dewatering on a filter press, mining and mineral processing return water.
Fine particles under 1 micron carry a strong negative surface charge that keeps them dispersed — they will not settle without charge neutralization first. These need a coagulant. If the resulting microflocs are large enough to remove by clarification or filtration without further bridging, a coagulant alone may be sufficient.
Common coagulant-only applications: low-turbidity raw water clarification, light oil and grease emulsions, color removal in lightly contaminated streams, some metal precipitation applications.
Most industrial wastewater needs both. Facilities discharging to a POTW under strict TSS, BOD, or metals permit limits typically cannot achieve compliant discharge with one chemistry alone. The coagulant neutralizes charges first; then the flocculant builds settleable flocs large enough for the separation step — DAF, clarifier, or filter press.
Common both-required applications: manufacturing process water with mixed contaminants, food processing wastewater with FOG and suspended organics, metal finishing and fabrication discharges, any stream where dissolved air flotation (DAF) is used.
The 5-minute Water Confidence Assessment maps your facility’s wastewater profile to the treatment gaps most likely driving your costs.
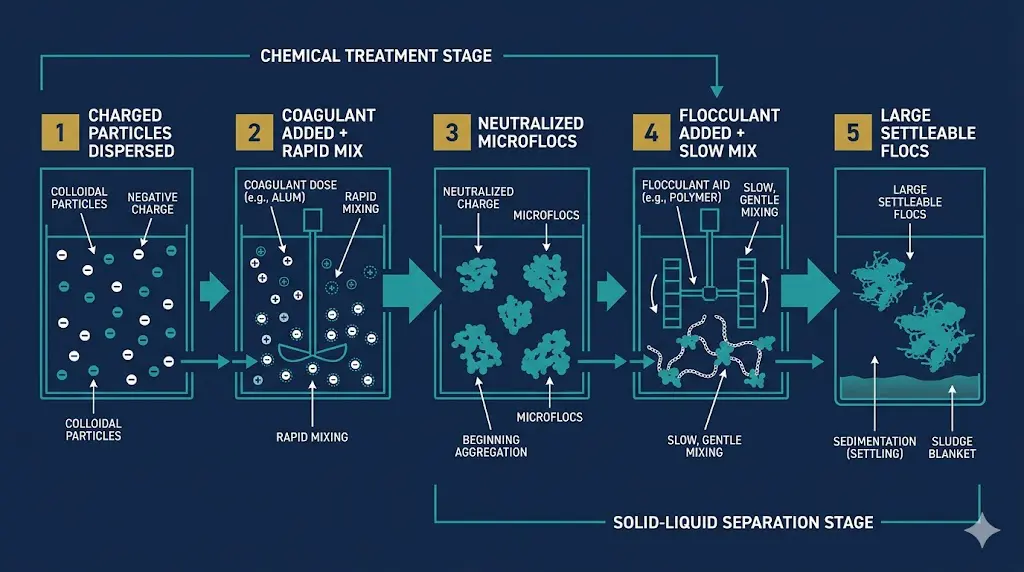
Once you know you need a flocculant, the next question is charge. Flocculants come in three charge types, and using the wrong one is one of the most common reasons polymer programs fail.
Cationic flocculants carry a positive charge and bind negatively charged particles. Most organic solids are negatively charged — sludge from biological treatment, FOG-laden wastewater from food processing or dairy operations, and most municipal-style waste streams. If you are dewatering sludge or treating food and beverage, dairy, or rendering wastewater, cationic is almost always the right answer.
Anionic flocculants carry a negative charge and bind positively charged particles. Mineral particles — after coagulation — tend to be positively charged, which makes anionic flocculants the dominant choice for stone wash, aggregate processing, mining, and mineral beneficiation water. These applications almost never need cationic.
Non-ionic flocculants have no net charge and rely on bridging alone. They work in high-chemistry environments where ionic interference would compromise a charged polymer, or in applications with mixed-charge solids where a cationic or anionic would bind some particles and repel others.
Within each charge category there are dozens of options varying by molecular weight and charge density. The right choice is rarely the same as your neighbor’s. ChemREADY’s FlocREADY line covers all three charge types in dry, gel, and emulsion forms, tuned to specific industrial profiles.
Common Mistake: Running cationic flocculant on mineral wash water, or anionic flocculant on organic sludge. The chemistry will not work, the dose will keep climbing, and the operator will assume the product is bad. The product is fine. The charge is wrong.
If your wastewater needs a coagulant, you also need to choose between inorganic and organic types. The trade-off is not about effectiveness — both work. It is about total program cost.
Inorganic coagulants (alum, ferric chloride, polyaluminum chloride / PAC) are inexpensive on a unit basis, work across a wide range of water chemistry profiles, and produce strong sweep-floc that captures organics and oils alongside the target solids. The trade-off: they generate 20–40% more sludge volume than organic coagulants, which raises your dewatering capacity requirements and disposal costs.
Organic coagulants (PolyDADMACs, polyamines) work through pure charge neutralization at much lower doses — typically 5–25 mg/L vs. 30–150 mg/L for inorganic types. They produce far less sludge, do not depress pH, and often deliver lower total program cost for facilities paying by the ton on sludge hauling. The trade-off: higher product cost per gallon and narrower performance windows on certain water profiles.
For most facilities, the right answer is not “which coagulant is cheaper per drum.” It is whichever one produces the lowest total cost when you add chemical spend, sludge volume, hauling fees, and disposal costs together. ChemREADY stocks both inorganic and organic coagulants because the right answer depends on the math at your specific facility.
We pull a sample, run the bench tests, and recommend the chemistry that performs in your stream.

Send us a sample — we'll run the analysis and translate it into plain-language recommendations for your treatment program.
Across hundreds of industrial facilities, the same three mistakes appear repeatedly. Each one is a fixable program problem, not a chemistry problem.
Running 30–50 mg/L of polymer where 5–8 mg/L would do the job. This happens when nobody runs jar tests, when dosing is “set and forget,” or when a charge mismatch makes the operator keep increasing the pump rate. Polymer overdose actually produces worse settling — excess polymer recoats the floc and re-stabilizes it, reversing the flocculation process entirely.
Adding flocculant before coagulant, or dosing both at the same injection point. The coagulation-flocculation process has a required order: coagulant first to neutralize charge, then flocculant to bridge. Mixing them at the same point — or reversing the sequence — destroys the mechanism. This is not a product failure. It is an installation problem.
Buying dry polymer for an operation that lacks the makedown equipment to hydrate it properly, or specifying emulsion polymer where dry would work cleanly. The product specification is correct. The physical form does not match the facility’s infrastructure. Poorly hydrated dry polymer behaves like fish eyes in the stream — visible chunks that never fully activate.
Each of these is identified and corrected in a structured facility review. ChemREADY’s Source-to-Discharge service covers a jar test program, observation of the makedown system in person, and program tuning against actual permit parameters. For most facilities, it pays for itself in the first quarter through reduced polymer spend and more consistent discharge compliance.
ChemREADY runs facility-level wastewater reviews tied to your actual permit parameters and discharge profile. We pull a sample, run the bench tests, and recommend the chemistry that performs in your stream.
Book a 30-minute conversation directly. We'll discuss your wastewater treatment situation and whether working together makes sense.
Book a 30-minute call →Take a look at our wastewater treatment services first. See where you stand across discharge compliance, effluent quality, and chemical treatment programs — then decide if a conversation makes sense.
Find out what a buyer would see →Browse everything we do in wastewater treatment — chemicals, equipment, and services — at your own pace, no conversation required.
Explore our wastewater solutions →We work with advisors who encounter infrastructure gaps in industrial wastewater client businesses. If you're looking for a referral partner for pre-sale readiness work, let's talk.
Learn about our partner program →